Problem framing
We were brought in to solve a business problem, reduce projected support costs of a Medical Device.
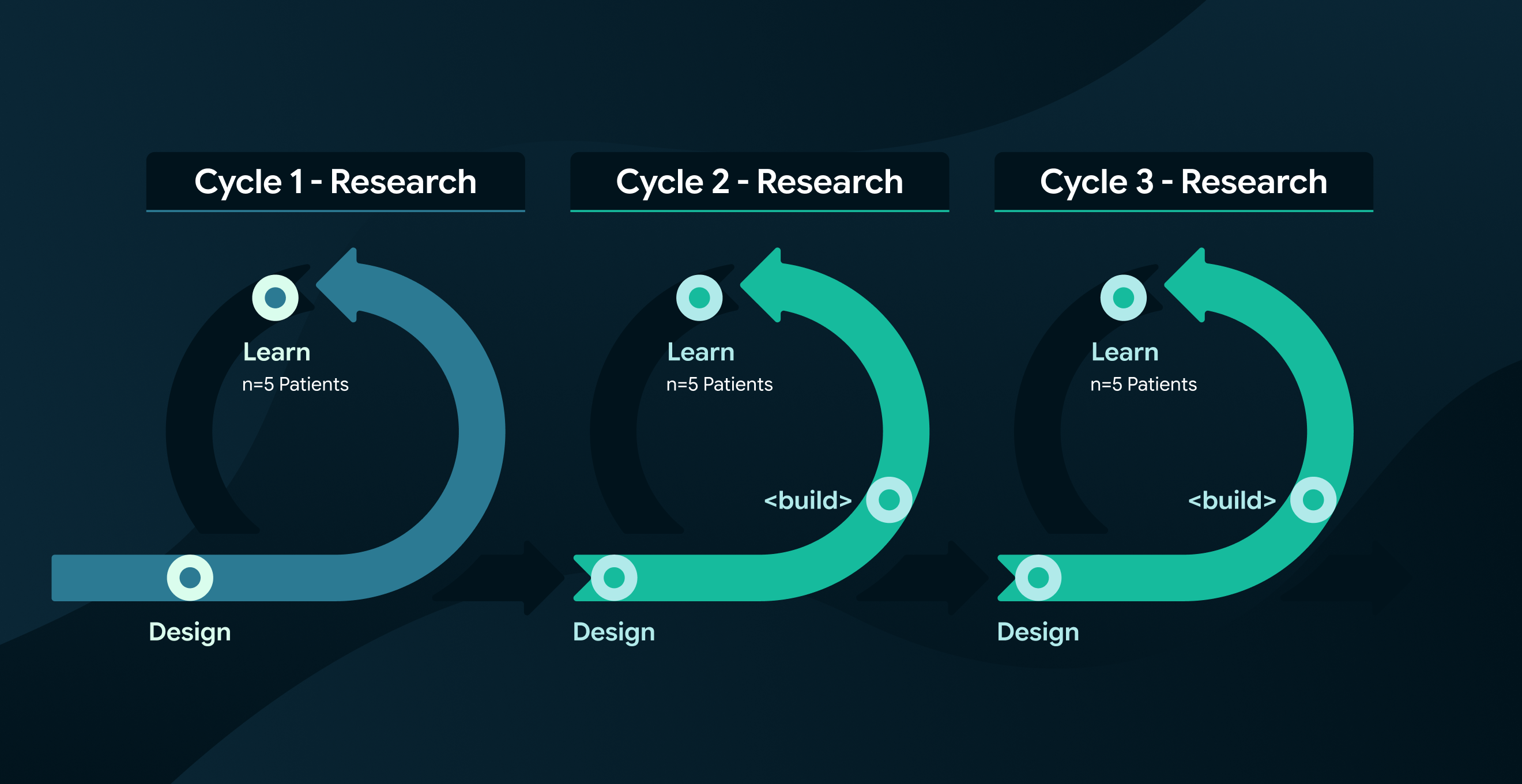
With very little budget for foundational research, we took a lean, iterative approach to the user problem, padding out our understanding of the problem space as we designed and tested.
The business problem
Research shows that our user group tend to rely on customer support to resolve issues with their medical device, creating escalating support costs as usage scales.

The user group's acessability issues
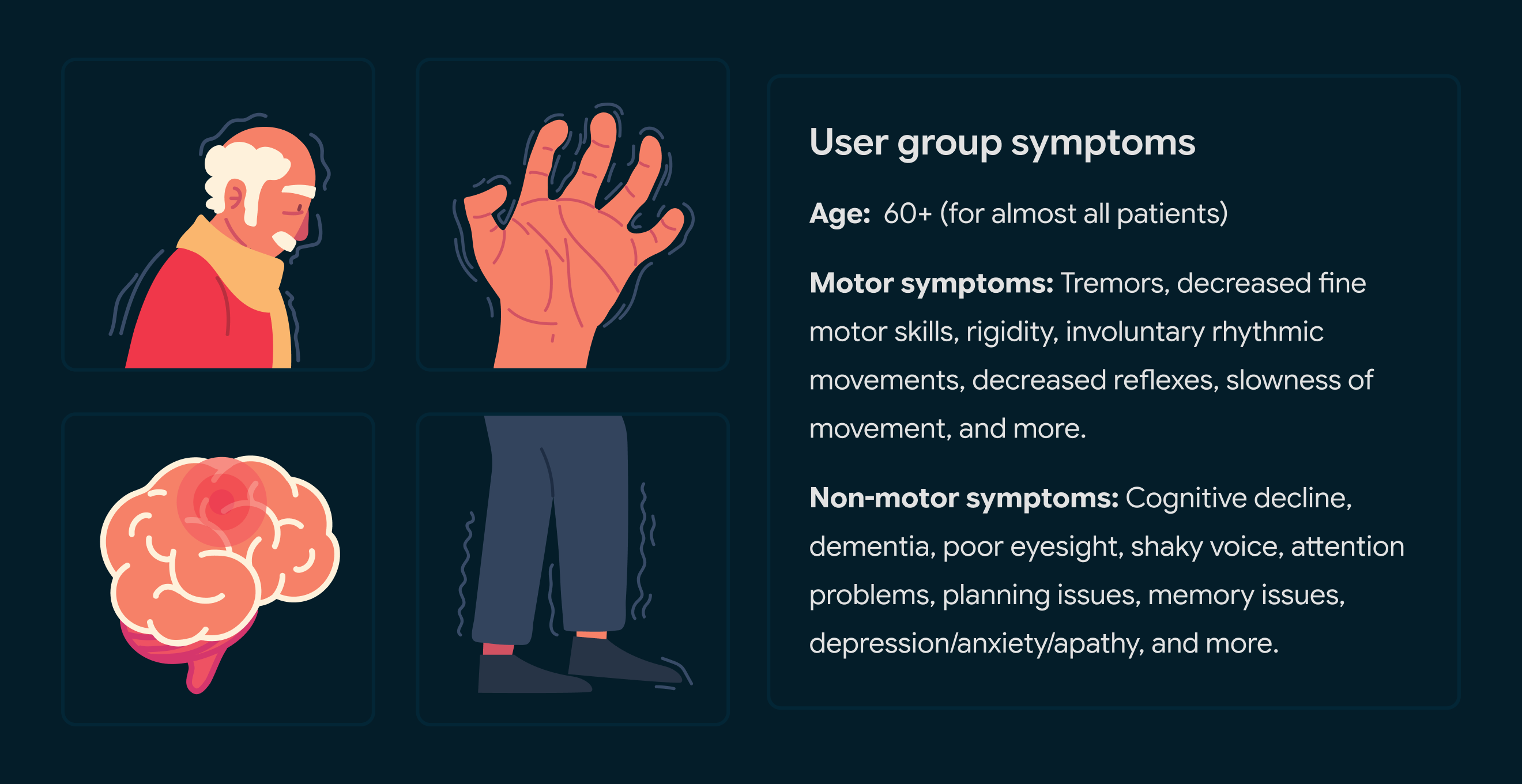
The Medical Device was designed specifically for a disabled user group with significant accessibility issues (exact patient group redacted online due to request from client).
It was essential to test and get feedback with them directly to ensure the product worked for their unique case.
Upfront contraints
Because this was for a regulated Medical Device, we had to surface content that had already passed regulatory.
“The regulatory consultant says we need to solve this in a safe and effective manner by surfacing user manual content that has already passed HF evaluation.” ~ Product Manager
User testing plan
With a budget for 15 in-person participants, we prioritised testing with the Medical Device in as near to a real-world context as possible to gather meaningful insight.
I worked closely with researchers, management, and the client to structure the work into three sprint-based testing rounds where we could maximise learning across each cycle.